二氢-FK 506
dihydro-fk 506
CAS: 104987-30-6
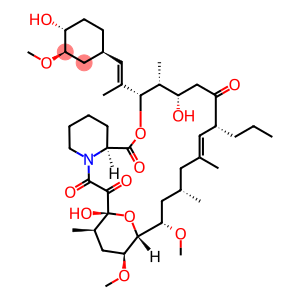
Molecular Formula: C44H71NO12
二氢-FK 506 - Names and Identifiers
| Name | dihydro-fk 506 |
| Synonyms | Dihydro-FK506 dihydro-fk 506 TsukubaMycin B Dihydro-FK506 DIHYDRO-FK506 TacroliMus 8-Propyl Analog (USP) Tacrolimus EP Impurity E (Dihydro Tacrolimus) 15,19-Epoxy-3H-pyrido[2,1-c][1,4]oxaazacyclotricosine-1,7,20,21(4H,23H)-tetrone, 5,6,8,11,12,13,14,15,16,17,18,19,24,25,26,26a-hexadecahydro-5,19-dihydroxy-3-[(1E)-2-[(1R,3R,4R)-4-hydroxy-3-methoxycyclohexyl]-1-methylethenyl]-14,16-dimethoxy-4,10,12,18-tetramethyl-8-propyl-, (3S,4R,5S,8R,9E,12S,14S,... |
| CAS | 104987-30-6 |
| EINECS | 806-466-9 |
| InChI | InChI=1S/C44H71NO12/c1-10-13-31-19-25(2)18-26(3)20-37(54-8)40-38(55-9)22-28(5)44(52,57-40)41(49)42(50)45-17-12-11-14-32(45)43(51)56-39(29(6)34(47)24-35(31)48)27(4)21-30-15-16-33(46)36(23-30)53-7/h19,21,26,28-34,36-40,46-47,52H,10-18,20,22-24H2,1-9H3/b25-19+,27-21+/t26-,28+,29+,30-,31+,32-,33+,34-,36+,37-,38-,39+,40+,44+/m0/s1 |
二氢-FK 506 - Physico-chemical Properties
| Molecular Formula | C44H71NO12 |
| Molar Mass | 806.03 |
| Density | 1.18±0.1 g/cm3(Predicted) |
| Melting Point | 87-94°C |
| Boling Point | 875.5±75.0 °C(Predicted) |
| Solubility | Soluble in DMSO |
| Appearance | White powder |
| pKa | 9.97±0.70(Predicted) |
| Storage Condition | 2-8℃ |
二氢-FK 506 - Risk and Safety
| HS Code | 2934990002 |
二氢-FK 506 - Introduction
Tacrolimus Impurity 6, also known as Tacrolimus EP Impurity E or Tacrolimus 8-Propylene Analog, is a chemical compound that is closely related to the immunosuppressant drug Tacrolimus (also known as FK506). It is categorized as an impurity, which means it is an unintended product or byproduct that can be found during the manufacturing process of Tacrolimus.
As an analog of Tacrolimus, Tacrolimus Impurity 6 shares a similar chemical structure but may have slightly different properties and effects. It is primarily used for analytical purposes in pharmaceutical research and development. Researchers utilize impurities like Tacrolimus Impurity 6 to identify, analyze, and quantify the impurities present in Tacrolimus formulations.
It is important to note that Tacrolimus Impurity 6 should not be used for human consumption or as a therapeutic agent, as its safety and efficacy have not been established for such purposes. Instead, its significance lies in its role as a reference standard, helping researchers ensure the quality, purity, and potency of Tacrolimus drugs on the market.
In summary, Tacrolimus Impurity 6 (Tacrolimus EP Impurity E or Tacrolimus 8-Propylene Analog) is an impurity commonly detected in Tacrolimus formulations. It serves as a reference standard for research purposes, allowing scientists to study and monitor impurities in order to ensure the quality of Tacrolimus medications.
Supplier List
CAS: 104987-30-6
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 104987-30-6
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 104987-30-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 104987-30-6
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
CAS: 104987-30-6
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 104987-30-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025


